Metals are elements that typically conduct electricity and heat, show metallic luster, deform plastically under stress, and give up electrons in chemical reactions; nonmetals usually lack metallic luster, are poor electrical conductors, tend to gain or share electrons, and form covalent or acidic oxides; metalloids show hybrid behavior, possessing a mixture of metallic and nonmetallic properties that make them valuable in semiconductors and specialized alloys.
Historical classification and rationale
Classification of elements into metals and nonmetals dates to early chemistry and mineralogy when properties like malleability, luster, and ability to form alloys were primary sorting features. Periodic table development refined the system: groups on the left and center are predominantly metallic, groups to the right are largely nonmetallic, with a narrow diagonal band of elements that display mixed features: the metalloids. Practical driving forces for classification included: metallurgical utility, ease of extraction, economic importance, and predictive chemical behavior. In modern practice, classification supports material selection, process design, and predictive modeling.
Defining properties: physical criteria
A technical checklist useful during identification:
-
Electrical conductivity: High in most metals due to a sea of delocalized electrons. Low in most nonmetals. Metalloids often exhibit semiconducting conductivity that changes strongly with temperature and doping.
-
Thermal conductivity: Metals typically conduct heat efficiently; nonmetals have poorer thermal conduction; metalloids vary.
-
Luster and reflectivity: Metals display characteristic metallic luster and high reflectivity on polished surfaces; many nonmetals are dull or transparent; metalloids often show a subdued metallic sheen.
-
Malleability and ductility: Metals deform plastically under stress permitting rolling, forging, or drawing; nonmetals are brittle in bulk solid form; metalloids may be brittle but can sometimes be worked under specialized conditions.
-
Mechanical strength: Metals span a large range from soft (e.g., sodium) to extremely strong (e.g., high-strength steels); mechanical performance depends heavily on microstructure and alloying.
-
Density: Metals generally have higher densities than nonmetals, although exceptions exist (e.g., dense nonmetal iodine).
-
Phase at STP (Standard Temperature and Pressure): Most metals are solid at STP (mercury an important liquid exception). Nonmetals may be gases (O₂, N₂), liquids (bromine), or solids (carbon, sulfur).
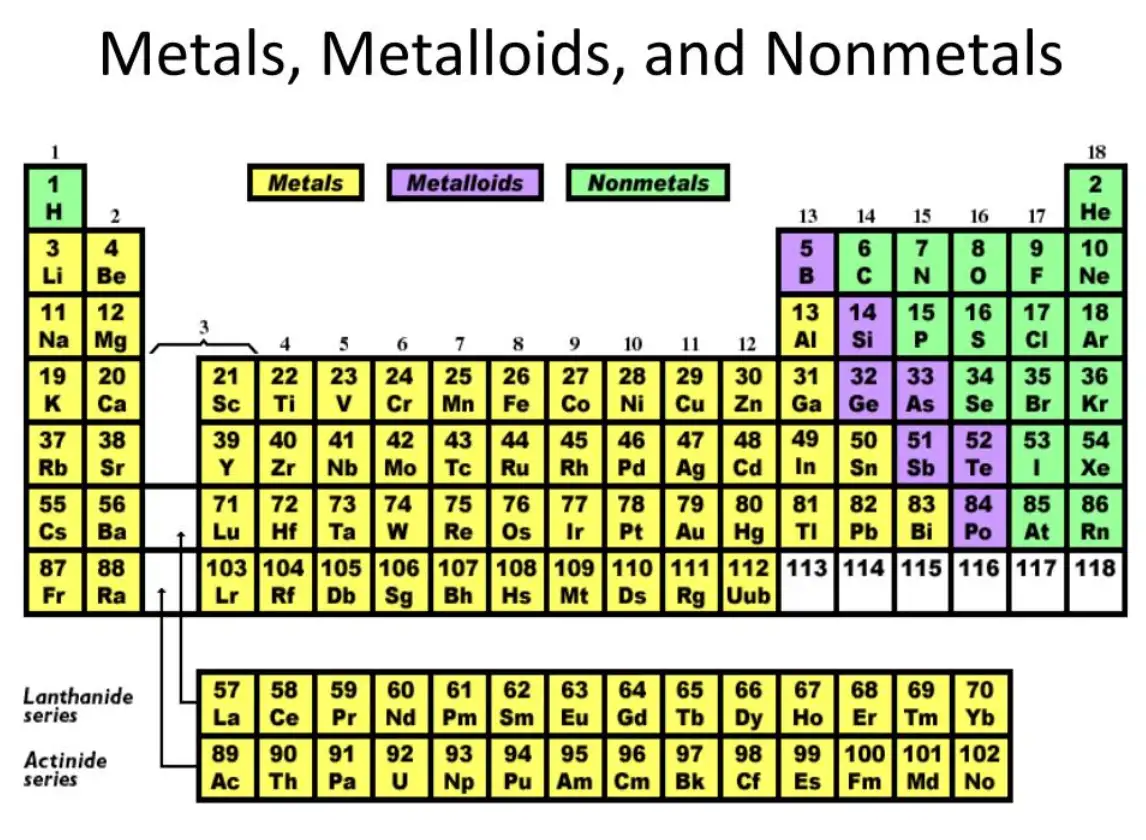
Defining properties: chemical behavior and bonding tendencies
Chemical differences arise from electronegativity, ionization energy, and electron affinity:
-
Tendency to lose electrons: Metals have relatively low ionization energies and form cations easily. They form ionic bonds with many nonmetals and metallic bonds in pure form or alloys.
-
Tendency to gain or share electrons: Nonmetals have higher electronegativities and ionization energies, favoring electron gain or sharing; they form covalent networks, diatomic molecules, or polyatomic anions.
-
Oxide character: Metal oxides tend to be basic or amphoteric, while nonmetal oxides are often acidic; metalloids may form amphoteric oxides that display both behaviors depending on the reacting partner.
-
Redox behavior in typical environments: Metals act as reducing agents in contact with acidic environments; nonmetals often oxidize metals or act as oxidizers where appropriate.
-
Complex formation: Transition metals form coordination complexes readily due to d-orbital participation; nonmetals and metalloids participate differently in complex chemistry.
Electronic structure and periodic placement
Understanding where each category appears on the periodic table clarifies both trends and exceptions:
-
Metals: Occupy the s-block, d-block, f-block, and part of the p-block. Characterized by partially filled conduction bands and low work functions. Transition metals (d-block) show variable oxidation states and complex magnetic and catalytic properties.
-
Nonmetals: Cluster toward the top-right of the periodic table (excluding noble gases in a separate category). Small atomic radii, high electronegativities, and filled or nearly filled valence shells produce strong directional bonding.
-
Metalloids: Straddle the stair-step between metals and nonmetals: boron, silicon, germanium, arsenic, antimony, tellurium, polonium (classifications vary). Their band structures create narrow band gaps suitable for semiconductor applications.
Metalloids: definition, identification, and borderline behavior
Metalloids merit special attention because their intermediate properties underpin modern electronics and speciality alloys:
-
Canonical metalloids: Boron (B), Silicon (Si), Germanium (Ge), Arsenic (As), Antimony (Sb), Tellurium (Te). Polonium sometimes included but has radioactive considerations.
-
Why metalloids matter: Their semiconducting behavior can be tuned by doping and temperature; they form glassy and ceramic phases with nonmetals; they function as alloying elements that modify mechanical and thermal properties.
-
Structural forms: Many metalloids form covalent networks (e.g., silicon) that are brittle yet electrically active when doped. Others (e.g., antimony) have layered crystal structures that show anisotropic behavior.
-
Industrial role: Semiconductors (Si, Ge), flame retardants and alloy modifiers (Sb), thermoelectric materials (Te), and specialty ceramics (B).
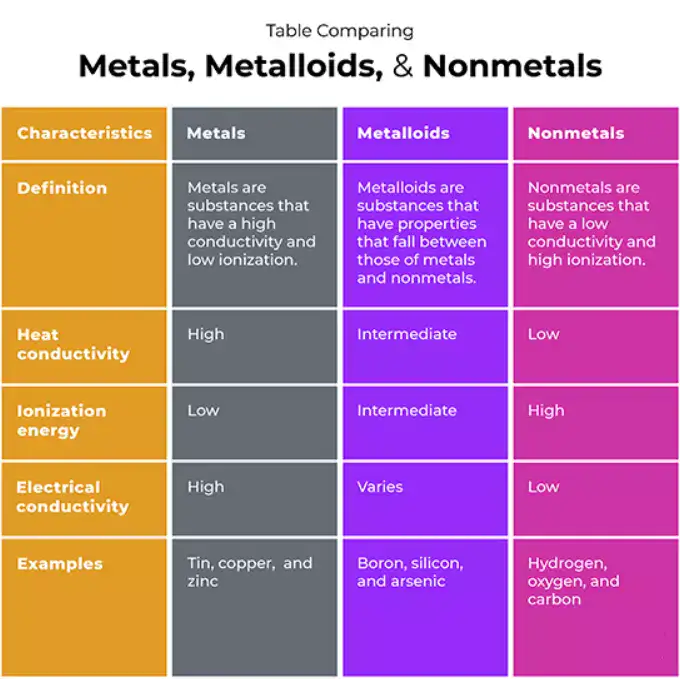
Comparative tables — quick reference
Table 1 — High-level physical and chemical comparison
| Property / Feature | Metals | Nonmetals | Metalloids |
|---|---|---|---|
| Typical electrical conductivity | High | Low to none | Intermediate / semiconducting |
| Thermal conductivity | High | Low | Moderate |
| Luster | Metallic (shiny) | Dull / nonmetallic | Often metallic sheen |
| Mechanical behavior | Ductile / malleable (many) | Brittle (most solids) | Brittle to semi-ductile |
| Common oxides | Basic / amphoteric | Acidic / covalent | Amphoteric |
| Typical bonding | Metallic / ionic | Covalent / molecular | Covalent with partial metallic character |
| Periodic location | Left / center / bottom | Right / top | Stair-step boundary |
Table 2 — Representative examples and typical uses
| Category | Representative elements | Typical industrial uses |
|---|---|---|
| Metals | Iron (Fe), Copper (Cu), Aluminum (Al), Nickel (Ni), Titanium (Ti) | Structural steel, electrical wiring, aerospace, corrosion-resistant alloys |
| Nonmetals | Oxygen (O), Nitrogen (N), Sulfur (S), Carbon (C), Phosphorus (P) | Chemical feedstocks, polymers, oxidizers, catalysts (carbon supports) |
| Metalloids | Silicon (Si), Germanium (Ge), Boron (B), Antimony (Sb), Tellurium (Te) | Semiconductors, thermoelectrics, special ceramics, alloy additives |
Table 3 — Electronic / bonding signature and extraction notes
| Element class | Valence electron behavior | Typical ore source | Typical extraction or processing route |
|---|---|---|---|
| Metals | Delocalized conduction electrons | Ores: oxides, sulfides | Pyrometallurgy, hydrometallurgy, electrolytic refining |
| Nonmetals | Electron localization, covalent networks | Native gases, mineral deposits (e.g., sulfur), organic sources | Chemical synthesis, gas separation, mining (e.g., graphite) |
| Metalloids | Narrow band gap, directional covalent bonds | Silicates, sulfides, native forms | Chemical purification (zone refining for Si), roasting and leaching |

Geological occurrence, extraction, and commercial forms
From a procurement and processing standpoint:
-
Metals: Frequently mined from oxides and sulfide minerals (e.g., hematite for iron, chalcopyrite for copper). Smelting removes oxygen or sulfur; electrorefining and magnetohydrometallurgical steps yield high-purity metal. Commercial forms: ingots, billets, sheet, coil, wire, powder.
-
Nonmetals: Occur in elemental, molecular, or mineral forms. Carbon appears as graphite or diamond; sulfur occurs in elemental deposits or volcanic emissions; nitrogen and oxygen are atmospheric gases obtained by air separation. Commercial forms include gases, powders, and molecular solids.
-
Metalloids: Silicon is extracted from quartz and refined to metallurgical-grade silicon via carbothermic reduction, then purified with chemical methods (e.g., the Siemens process or fluidized-bed reactors) for electronic-grade Si. Boron is sourced from borates; germanium often recovered from zinc ores or coal fly ash.
Industrial uses and examples with practical procurement notes
Element-by-element summaries with buying considerations:
-
Iron / Steel (metal): Primary structural metal. Purchase specifications usually reference standards (ASTM A36, A992, etc.), and microstructure matters (e.g., ferritic, martensitic). Corrosion resistance and weldability depend on composition and heat treatment.
-
Copper (metal): Critical for electrical conduction. Purchase grades (e.g., C11000, C10200) indicate purity and conductivity. Oxygen-free copper used where high conductivity and low impurity levels are required.
-
Aluminum (metal): Preferred where low density and corrosion resistance are vital. Alloy series (1000 through 7000) define key properties. Surface finish and temper matter for forming vs. structural applications.
-
Silicon (metalloid): Electronic-grade silicon used for wafers; metallurgical-grade silicon used for alloys and silicones. Purchasing requires careful attention to purity (ppb levels for electronics).
-
Carbon (nonmetal): Graphite used as a high-temperature electrode and lubricant; activated carbon for adsorption. Forms and porosity govern performance.
Procurement tip: Request material certificates (mill test reports, chemical and mechanical test reports), confirm trace impurity limits for critical applications, and verify compliance with applicable standards (ASTM, ISO, EN).
Alloying, treatment, and property modification
How differences map to commonly used industrial processes:
-
Alloying metals: Incorporating elements changes electron density, lattice distortion, and phase stability. For example, carbon in iron forms steel with dramatically increased strength; chromium adds corrosion resistance in stainless steels.
-
Doping metalloids: Controlled introduction of donor or acceptor impurities converts intrinsic semiconductors into n-type or p-type, which dictates electrical behavior. Temperature and concentration regulate carrier mobility.
-
Nonmetal functionalization: Nonmetals form functional compounds (e.g., carbon in polymers, sulfur in vulcanized rubber) that deliver specific chemical or mechanical performance.
-
Heat treatment: Metals respond well to annealing, quenching, tempering, and age-hardening. Metalloids and nonmetallic materials require specialized thermal or chemical routes for property tuning (e.g., ceramic sintering, glass annealing).
Safety, handling, and environmental considerations
Practical safety points for each class:
-
Metals: Many metals produce toxic dust or fumes during cutting, welding, or smelting (e.g., lead, cadmium). Corroded metals can produce hazardous byproducts. Use appropriate PPE, ventilation, and meet waste disposal rules.
-
Nonmetals: Reactive nonmetals (e.g., phosphorus, chlorine gas) pose acute hazards; powders (e.g., carbon black) can present dust explosion risks. Follow MSDS guidance.
-
Metalloids: Some metalloids have toxicity concerns (e.g., arsenic compounds). Semiconducting grade materials may require contamination control and clean-room handling.
-
Environmental: Lifecycle impacts differ dramatically; metals often entail energy-intensive extraction, while some nonmetal production (like petrochemical-derived polymers) carries different pollutant profiles. Recycling and substitution considerations play a major role in procurement strategy.
How to identify an unknown sample in the lab or workshop
A stepwise practical protocol:
-
Visual inspection: Note color, luster, and texture. Metallic sheen suggests metal or metalloid.
-
Density measurement: Bulk density measured by displacement gives a strong clue. High density suggests metal (but check for heavy nonmetals like iodine).
-
Electrical test: Use a multimeter to measure DC conductivity. High conductivity indicates metal; low suggests nonmetal; temperature-dependent intermediate behavior suggests metalloid/semiconductor.
-
Mechanical test: Simple bend or scratch tests indicate ductility vs. brittleness. Metals typically deform; nonmetals often fracture.
-
Chemical spot tests: Reacting small samples with acids or using flame tests can identify class behavior—metals may dissolve to form colored ions; nonmetals produce gases or insoluble residues. Always follow safety protocols.
-
Spectroscopic confirmation: XRF, AES, ICP-MS, or XRD provide definitive composition and phase identification for procurement-grade verification.
FAQs
Q1: What fundamental electronic difference separates metals from nonmetals?
A: Metals possess delocalized valence electrons that form conduction bands, enabling free-electron movement under an electric field. Nonmetals have localized electrons occupying filled or nearly filled valence shells, supporting directional covalent bonds and resulting in poor electrical conduction in bulk.
Q2: Are metalloids always semiconductors?
A: Not always. Many metalloids have intrinsic semiconducting behavior with narrow band gaps (example: silicon, germanium), but physical form, doping, and temperature strongly influence conductivity; some metalloids behave more metallic in particular crystalline morphologies.
Q3: How do oxide properties reflect element classification?
A: Metal oxides are typically basic or amphoteric, neutralizing acids to form salts. Nonmetal oxides are usually acidic, forming acids in water (e.g., SO₂ → sulfurous acid). Metalloids commonly form amphoteric oxides with both basic and acidic reactions.
Q4: Which tests quickly determine if a metal is a transition metal or main-group metal?
A: Transition metals typically show multiple stable oxidation states, colored ions in solution, and complex formation behavior; main-group metals usually show fewer oxidation states and simpler binary compounds.
Q5: How important is purity for industrial silicon used in electronics?
A: Extremely important. Electronic-grade silicon requires impurity levels in the parts-per-billion (ppb) range or lower; trace metals can dramatically alter carrier lifetime and device yield.
Q6: Can nonmetals form alloys?
A: Not in the classical metallic sense. However, nonmetallic elements combine to form mixtures and composites (e.g., carbon in graphite-reinforced composites or polymer blends) that perform alloy-like roles in certain applications.
Q7: Which class corrodes most readily and how is protection achieved?
A: Corrosion propensity depends on environment and element: iron rusts readily in moist oxygen; many nonmetals do not corrode but can oxidize or decompose. Protection methods include coatings, cathodic protection, alloying, and environmental control.
Q8: Are there elements that defy simple classification?
A: Yes. Several elements lie near the metalloid boundary and show polymorphism with both metallic and nonmetallic phases. Additionally, elements under extreme pressure or temperature may switch behavior.
Q9: How do alloying additions of metalloids influence metal properties?
A: Small additions of metalloids (e.g., silicon, boron) refine grain size, alter hardenability, improve strength at temperature, or change electrical behavior. For example, silicon in steel influences deoxidation and magnetic properties.
Q10: What material standards should be consulted when specifying metals for procurement?
A: Common standards include ASTM (American), EN (European), ISO, JIS (Japanese) depending on region. For electronic materials, JEDEC and semiconductor-specific standards apply. Always request relevant mill test reports and compliance documents.
Authoritative references
- IUPAC — Periodic Table and Element Information
- NIST Chemistry WebBook — Thermochemical and Spectroscopic Data
- Royal Society of Chemistry — Interactive Periodic Table and Element Data
- USGS — Metals and Minerals Information (National Minerals Information Center)
Closing notes and recommended next steps
-
For procurement documentation, pair this conceptual material with applicable ASTM/EN standards for each specific metal or alloy.
-
For people preparing teaching materials or web content, transform the tables into responsive HTML with microdata for richer search engine results.
-
If further detail is needed on any single element, alloy system, or processing route (for example, heat-treatment schedules for specific steels, silicon wafer purity pathways, or corrosion test data), provide the element or alloy name and the intended application; a targeted technical brief can then be prepared.

