Sulfide stress corrosion cracking (commonly abbreviated SSC or SSCC) is a hydrogen-assisted brittle failure that occurs when susceptible steels and alloys are exposed to wet hydrogen sulfide (H₂S) under tensile stress. The phenomenon is controlled by three linked elements — a sour (H₂S-containing) environment that produces atomic hydrogen at the metal surface, tensile stress (residual or applied), and a microstructure / hardness that allows hydrogen-induced embrittlement and crack propagation. The most reliable ways to manage SSC are (1) select materials and hardness levels proven resistant under recognized standards (e.g., NACE TM0177, NACE MR0175 / ISO 15156), (2) control stresses by design and post-weld heat treatment, and (3) apply environmental controls and inspection programs. When these measures are combined, SSC risk can be reduced to acceptable levels for oil & gas, refining and related industries.
What is Sulfide Stress Corrosion Cracking?
Sulfide stress corrosion cracking is a hydrogen-related brittle cracking that happens in metallic components exposed to aqueous environments containing hydrogen sulfide (H₂S) and moisture while under tensile stress. Cracks are usually transgranular or intergranular and can cause sudden, catastrophic failure with little to no warning if not managed. Because H₂S is common in many oil, gas and refining streams, SSC is a central safety and reliability concern in hydrocarbon production, transportation and processing.
A short historical perspective
The oil and gas industry began documenting SSC in the 1950s–1960s as production moved into “sour” reservoirs containing H₂S. Early field failures prompted the development of practical laboratory test methods and eventually formal standards. NACE (now part of AMPP) codified test procedures (TM0177) and material recommendations (MR0175 / ISO 15156), which remain the backbone of industry practice for qualifying materials for sour service.
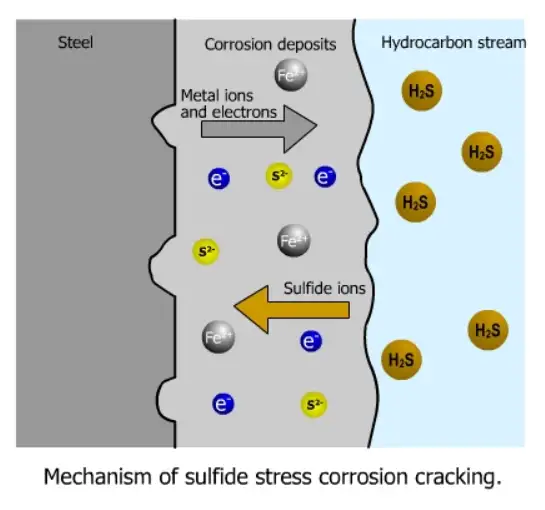
Mechanisms — how cracks start and grow
Contemporary research indicates SSC is not a single mechanism but a combination of related processes:
-
Hydrogen embrittlement / hydrogen-assisted cracking: H₂S in aqueous solution is reduced at the steel surface and, through cathodic reactions, generates adsorbed and atomic hydrogen. Atomic hydrogen diffuses into the metal, concentrates at trap sites (inclusions, dislocations, grain boundaries), and reduces ductility and fracture toughness. Fracture then occurs under tensile stress.
-
Stress corrosion cracking (SCC) contribution: In some situations anodic dissolution at the crack tip combines with hydrogen embrittlement to assist crack propagation. The relative contribution of anodic dissolution vs hydrogen embrittlement can vary with alloy, environment and stress state.
-
Critical factors: Crack initiation/propagation requires (1) availability of atomic H, (2) a stress intensity above local thresholds (often residual or weld-related stresses), and (3) a metallurgical condition (e.g., hardness, martensite, tempered martensite embrittlement) that enables brittle fracture at the relevant temperature and strain rate.
Environmental drivers that change SSC risk
Key environmental variables (practical interpretation):
-
H₂S partial pressure (or concentration): Higher H₂S tends to increase available hydrogen at the metal surface; standards and qualification schemes use H₂S partial pressure as a primary classification factor.
-
pH: Very low pH increases acidic dissolution; near-neutral pH “wet H₂S” conditions are particularly insidious because they allow sulfide formation with hydrogen generation.
-
Temperature: Increasing temperature can accelerate reaction rates and hydrogen diffusion but also change solubility—risk assessment must be temperature-specific.
-
Salinity and other ions (Cl⁻, CO₂): Chloride ions and CO₂ can worsen general corrosion and change hydrogen generation/diffusion behavior; mixed environments (H₂S + CO₂) need special evaluation.
-
Flow regime and oxygen: Flow affects surface film stability and oxygen presence can shift corrosion mechanisms away from the typical H₂S-only behavior.
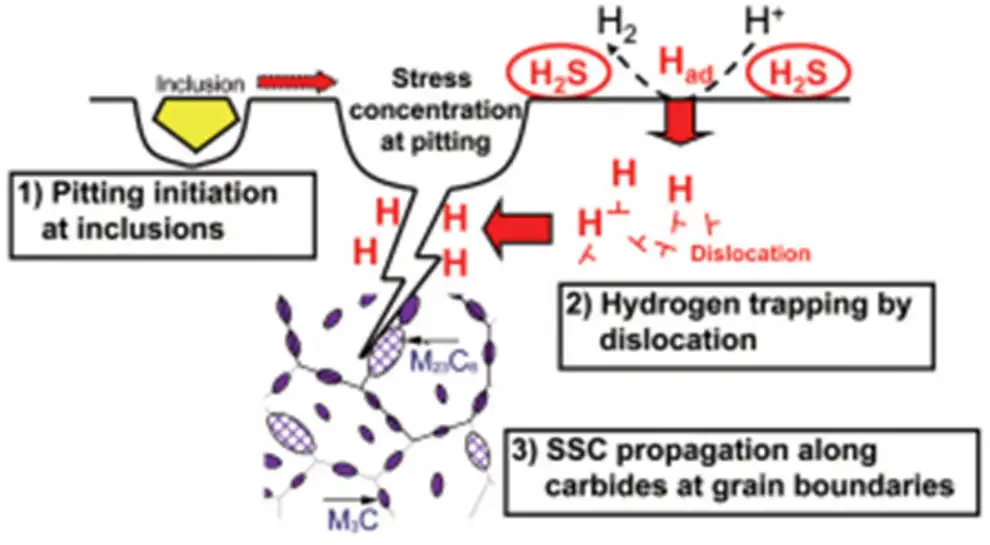
Which materials are susceptible — metallurgy matters
General rules (practical guidance):
-
Low-strength, low-hardness carbon steels can be susceptible to hydrogen-induced phenomena such as hydrogen-induced cracking (HIC) and stepwise cracking, but they tend to be less prone to SSC if hardness and yield strength are kept low.
-
High-strength steels and quenched & tempered steels (higher yield strength grades, e.g., API X70+ or high-strength line pipe and fasteners) are much more susceptible to SSC because their microstructure traps hydrogen and has lower tolerance for hydrogen embrittlement. Control of hardness is critical.
-
Austenitic stainless steels are not immune — under some wet H₂S conditions they can suffer hydrogen-assisted cracking or sulfide-related localized failures—so stainless grades must be evaluated per MR0175/ISO15156 guidance.
-
Nickel-base and other corrosion resistant alloys (CRAs) generally have superior resistance and are specified where SSC risk or performance requirements dictate, but they are costly and must be selected per standard limits and laboratory qualification.
Hardness guidance (rule of thumb): Many codes limit Brinell hardness (HB) for sour service; for carbon and low-alloy steels a typical upper limit used in specifications is about 197 HB (≈ Rc 22), while high-strength steels often require lower hardness in weld HAZ or heat-affected zones. Always follow the purchaser’s material spec and NACE/ISO limits for the specific environment.
Welding, fabrication and microstructures — special attention required
Weld heat-affected zones (HAZ), coarse grain regions, untempered martensite and quench microstructures are common hydrogen traps and crack initiation sites. Key fabrication controls:
-
Preheat and interpass temperature control to avoid formation of untempered martensite.
-
Post-weld heat treatment (PWHT) to temper martensitic structures and reduce residual stresses where applicable.
-
Hydrogen control in welding consumables (low hydrogen electrodes, proper storage and baking).
-
Hardness measurement of HAZ and welds to verify compliance.
Standards and test methods
The two most widely referenced documents are:
-
ISO 15156 / NACE MR0175 — establishes principles and specific rules for material selection for H₂S environments in oil & gas; it defines environmental envelopes and alloy classes and gives practical limits (for example, hardness limits, allowable alloys for specific H₂S partial pressures and temperatures).
-
NACE TM0177 (Sour Test Methods) — prescribes laboratory test procedures (tensile, bent-beam, C-ring, double cantilever beam) to evaluate SSC/SSCC resistance and is the de facto test reference for qualification. The standard describes solutions and test setups used to reproduce sour conditions in the laboratory.
Other useful documents include NACE MR0103 (refinery service) and various API and company material specifications that reference or supplement ISO/NACE guidance.
Laboratory testing — what the main methods tell you
Common SSC test methods (short summary):
| Test method | What it simulates | Typical result metric |
|---|---|---|
| Tensile (TM0177 Method A) | Constant tensile load in sour solution | Time-to-fail / fracture appearance. |
| Bent-beam (TM0177 Method B) | Bending stress + environment to accelerate cracking | Pass/fail after exposure period. |
| C-ring (TM0177 Method C) | Localized tensile stress around ring notch | Evaluation of crack initiation and propagation. |
| Double cantilever beam (TM0177 Method D) | Fatigue/crack growth under sour environment | Crack growth / fracture mechanics insight. |
Laboratory testing is necessary to qualify materials for specific sour environments but must be interpreted carefully because lab solutions and imposed stresses are simplifications of field conditions.
Inspection and monitoring — practical NDT and field controls
Because SSC can produce minimal external corrosion signatures before brittle failure, inspection programs should combine:
-
Regular ultrasound (UT) and phased-array UT to detect internal cracks and planar defects.
-
Magnetic particle testing (MPI) for surface/near-surface cracking on accessible ferrous components.
-
Hardness surveys across base metal, welds and HAZ to ensure compliance with specified limits.
-
Chemical monitoring of produced fluids and pipelines for H₂S concentration, pH and chloride levels.
-
Strain and stress monitoring in high-risk components to detect unusual loading or creep.
Proactive NDT frequency should be risk-based (higher for high-stress or high H₂S partial pressure locations).
Design and specification controls that reduce SSC risk
Key, high-impact specification items:
-
Material selection per ISO 15156 / NACE MR0175 with explicit environmental envelope (H₂S partial pressure, temperature, pH).
-
Hardness limits (Brinell or Rockwell) for base metal and weld HAZ. Typical supplier specs mandate HB ≤ 197 for many carbon steels in sour service, but values vary by alloy and service envelope.
-
Reduce tensile stresses by thicker sections, design geometry changes or use of post-tension relief (PWHT, stress-relief anneal).
-
Specify weld procedures and consumables that limit hydrogen pickup, and require PWHT where necessary.
Mitigation and operational controls
A layered approach produces best outcomes:
-
Material substitution: where practicable, use CRAs or nickel-base alloys for components exposed to the most aggressive H₂S conditions.
-
Hardness control: verify and enforce maximum hardness values for all procured and fabricated items.
-
PWHT and stress relief: use heat treatment to reduce residual tensile stresses and temper martensite.
-
Coatings and linings: protect surfaces from contact with sour aqueous films. Note: coatings can fail and hide corrosion—inspection access must remain.
-
Environmental control: remove or reduce dissolved H₂S by gas treatment, use of inhibitors or drying where possible.
-
Cathodic protection (CP): CP can reduce corrosion but must be applied carefully because it can change hydrogen evolution characteristics; CP does not remove the need for material selection limits.
Typical failure patterns and case notes
-
Weld HAZ cracking near girth welds: frequent when PWHT was omitted and hardness is high.
-
High-strength fastener failures: bolts in sour service have fractured unexpectedly due to combined stress and hydrogen embrittlement.
-
Line pipe longitudinal cracking: transgranular cracks initiating at surface defects or internal corrosion features in high-strength line pipe exposed to wet H₂S.
These examples emphasize the need to control both material/hardness and stress state.
Quick reference tables
Table A — Relative susceptibility by material family (practical view)
| Material family | Relative SSC susceptibility | Practical note |
|---|---|---|
| Low-carbon, annealed steels (low hardness) | Low–moderate | Hardness control critical. |
| Quenched & tempered high-strength steels | High | Avoid in sour service unless tested and controlled. |
| Martensitic stainless steels | Moderate–high | Tempering/PWHT and hardness control required. |
| Austenitic stainless steels | Low–moderate | Environment and cold work influence risk. |
| Corrosion-resistant alloys (nickel base, high-Ni CRAs) | Low | Costly but highly reliable in many sour envelopes. |
Table B — Common test methods and what they measure
| Test | Main use | Standard reference |
|---|---|---|
| Tensile in sour solution | Fail/no-fail under sustained tensile load | NACE TM0177 |
| Bent-beam | Susceptibility under bending stress | NACE TM0177 |
| C-ring | Local notch sensitivity | NACE TM0177 |
| HIC and SSC combined tests | Hydrogen blisters and crack sensitivity | NACE and ISO test recommendations |
FAQs
Q1: Is SSC the same as general stress corrosion cracking?
A: SSC is a specific hydrogen-assisted form of SCC that occurs in wet H₂S environments; it often involves hydrogen embrittlement mechanisms in addition to corrosion processes.
Q2: Which standard should I follow for material selection in sour oil & gas service?
A: ISO 15156 / NACE MR0175 is the primary international standard for selection and qualification of materials in H₂S service; NACE TM0177 provides the laboratory test methods.
Q3: Can coatings eliminate SSC risk?
A: Coatings reduce exposure but cannot be relied on as the only mitigation because coating damage or disbondment can expose the substrate under stress; combine coatings with appropriate materials and inspection.
Q4: What hardness limit should I specify?
A: There is no single limit for every case; many specs use ~197 HB (≈ Rc22) as a reference for carbon steels, but the precise limit must come from the standard/engineering assessment for the specific H₂S partial pressure and temperature.
Q5: Are stainless steels immune?
A: No — some stainless steels may be susceptible in specific H₂S conditions or if cold-worked; always validate selection against ISO/NACE guidance.
Q6: Do cathodic protection systems prevent SSC?
A: CP controls corrosion but does not eliminate hydrogen generation from sour environments and, if improperly applied, can change hydrogen evolution; CP is a supplementary control, not a replacement for appropriate materials and hardness limits.
Q7: Is lab testing reliable for field performance?
A: Lab tests are essential for qualification but must be interpreted with caution — replicate as many field conditions as possible and use conservative acceptance criteria.
Q8: What inspection frequency is recommended?
A: Inspection should be risk-based: higher H₂S partial pressure, higher stress, or prior failures require more frequent inspection. NDT types should match expected failure modes (UT, MPI, hardness checks).
Q9: What are early warning signs of SSC?
A: Often there are few external signs; look for stress-related distortion, new leaks, or changes in acoustic emission/strain data. Regular NDT is the best early detection.
Q10: If a component fails by SSC, what next steps should be taken?
A: Immediate shutdown and containment, metallurgical failure analysis, review of material/hardness records, reassessment of environment and stresses, and implement corrective specification and inspection changes before restart.
Short checklist for procurement and engineering specifications
-
Quote ISO 15156 / NACE MR0175 compliance and specify the environmental envelope (H₂S partial pressure, pH, temperature).
-
Require NACE TM0177 test evidence for nonstandard or high-risk materials.
-
Specify maximum hardness values for base metal, welds and HAZ and require hardness maps.
-
Insist on weld procedure qualification (PQR/WPS) with low hydrogen consumables and PWHT where indicated.
-
Define NDT/inspection plan and risk-based monitoring intervals.
Closing
Sulfide stress corrosion cracking remains a principal integrity threat in sour hydrocarbon environments. The combination of well-developed industry standards (ISO 15156 / NACE MR0175 and NACE TM0177), engineering controls (hardness limits, PWHT, material substitution), and disciplined field inspection programs provides a robust framework to prevent catastrophic SSC failures. Continuous monitoring of research (hydrogen diffusion, new CRAs, advanced NDT) and rigorous application of test data to real field envelopes are essential for safe, reliable operation.
Authoritative references
- ISO 15156: Petroleum and natural gas industries — Materials for use in H₂S-containing environments (ISO)
- Evaluation of Stress Corrosion Cracking, Sulfide (AMPP / Corrosion Journal)
- Hydrogen-induced cracking and blistering in steels — Review (NIST / ML Martin)
- Corrosion-Resistant Alloys in Oil & Gas — Selection Guidelines (Nickel Institute)

